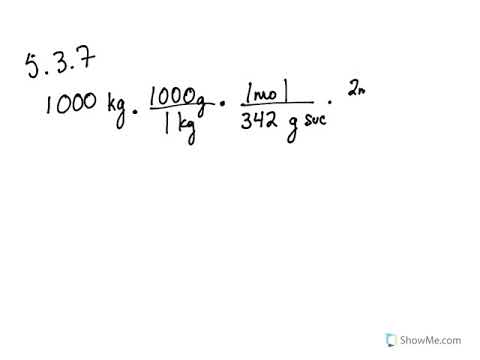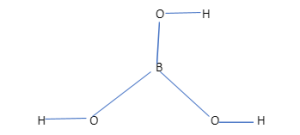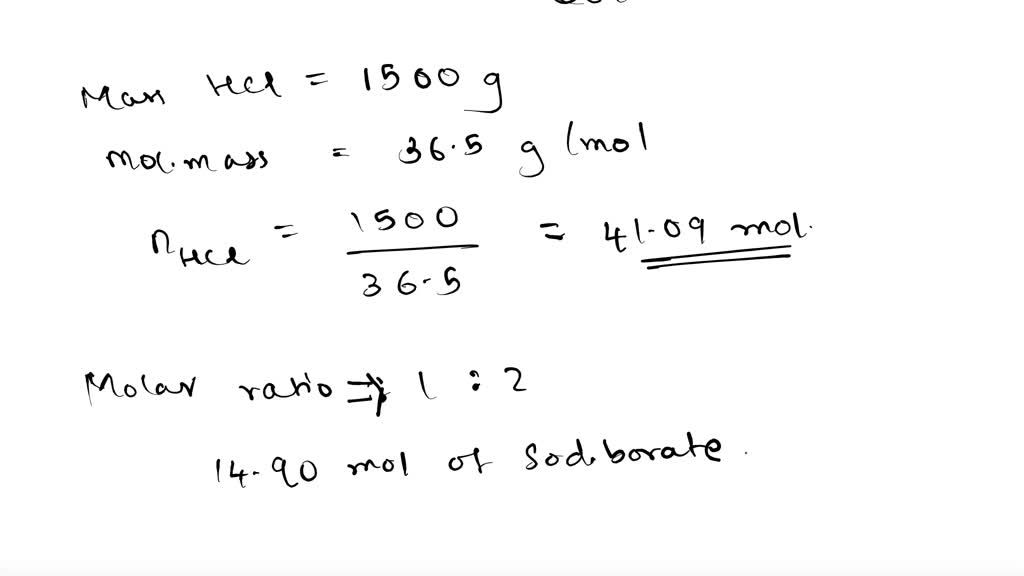
SOLVED: The raw materials, 3 kg of sodium borate along with 1.5 kg of hydrochloric acid will need to produce boric acid. Show the balanced chemical reaction of sodium borate plus hydrochloric

Ultralong Room‐Temperature Phosphorescence from Boric Acid - Zheng - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Calculate the mass of 1.00 mmol (S) of trans-cinnamic acid, and the theoretical yield of 2,3-dibromo-3phenylpropanoic acid. | Homework.Study.com

Boric Acid, a Lewis Acid With Unique and Unusual Properties: Formulation Implications - Journal of Pharmaceutical Sciences

SOLVED: Boron (B), one of the constituent elements of boric acid, naturally occurs as isotopes of 10B and 11B with percent natural abundance as 20% and 80%, respectively. Boric acid (H3BO3) is

Crosslinking Mechanism of Boric Acid with Diols Revisited - Rietjens - 2005 - European Journal of Inorganic Chemistry - Wiley Online Library